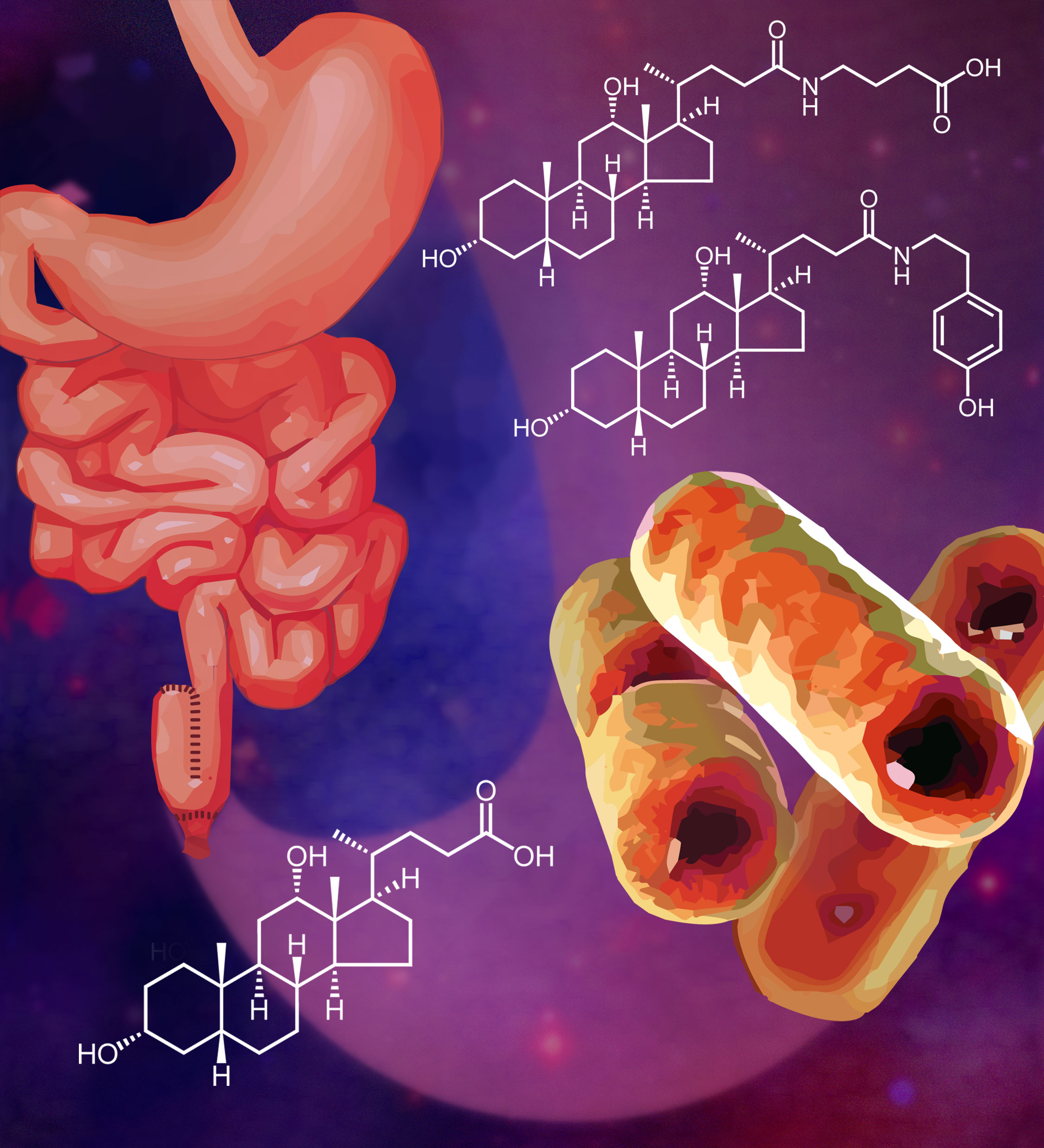
- Cover image artwork for Mullowney, M. W.; Fiebig, A.; Schnizlein, M. K.; McMillin, M.; Koval, J.; Rubin, D.; Dalal, S.; Sogin, M.L.; Chang, E. B.; Sidebottom, A. M.; Crosson, S. Microbially-catalyzed conjugation of GABA and tyramine to bile acids. J. Bacteriol. 2024, 206 , e00426–23.
- Animated version of cover image artwork for McGee, J.P.; Melani, R.D.; Yip, P.F.; Senko, M.W.; Compton, P.D.; Kafader, J.O.;Kelleher, N.L. Isotopic Resolution of Protein Complexes up to 466 kDa Using Individual Ion Mass Spectrometry. Anal. Chem. 2020. 93, 2723-2727.
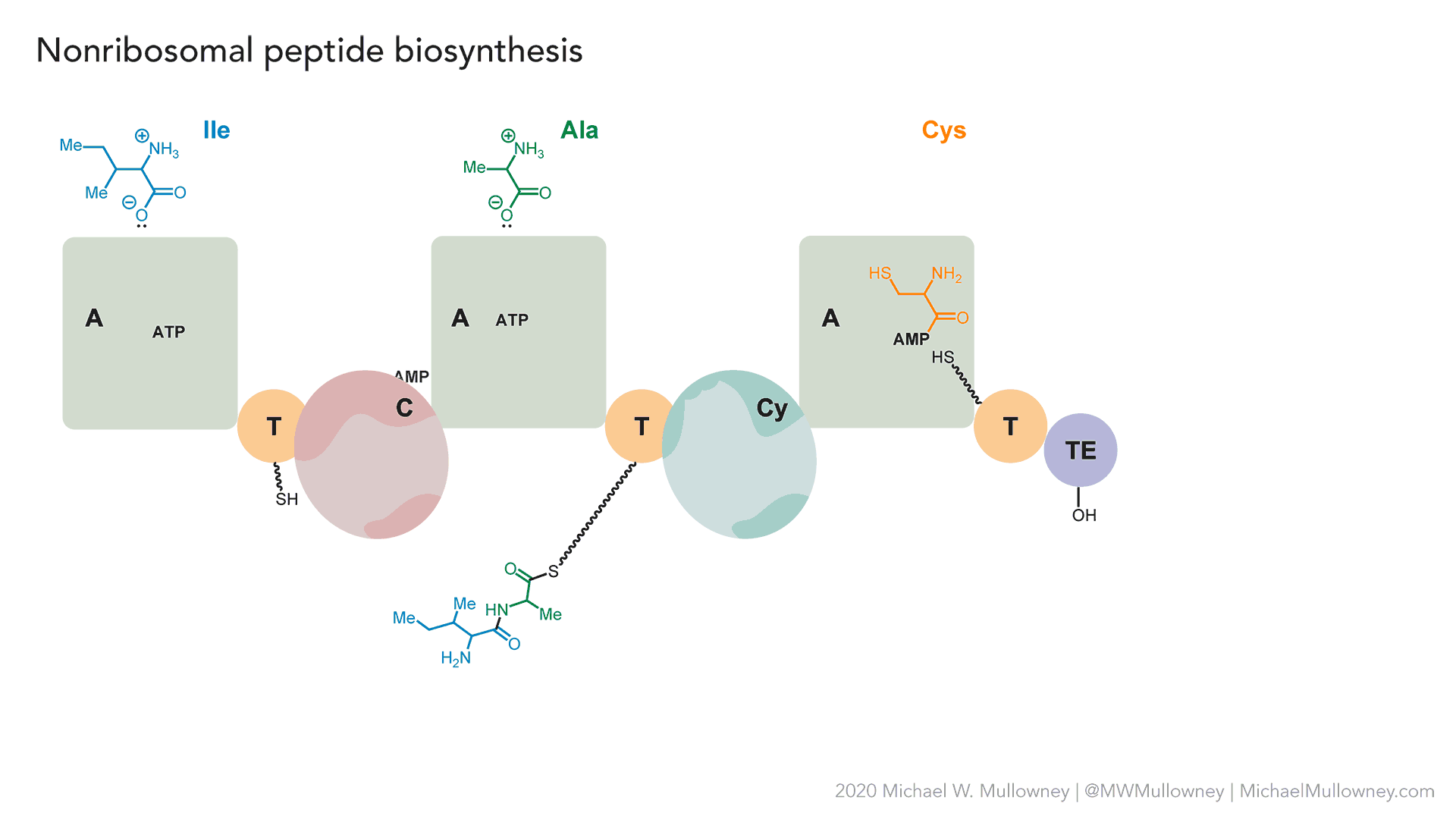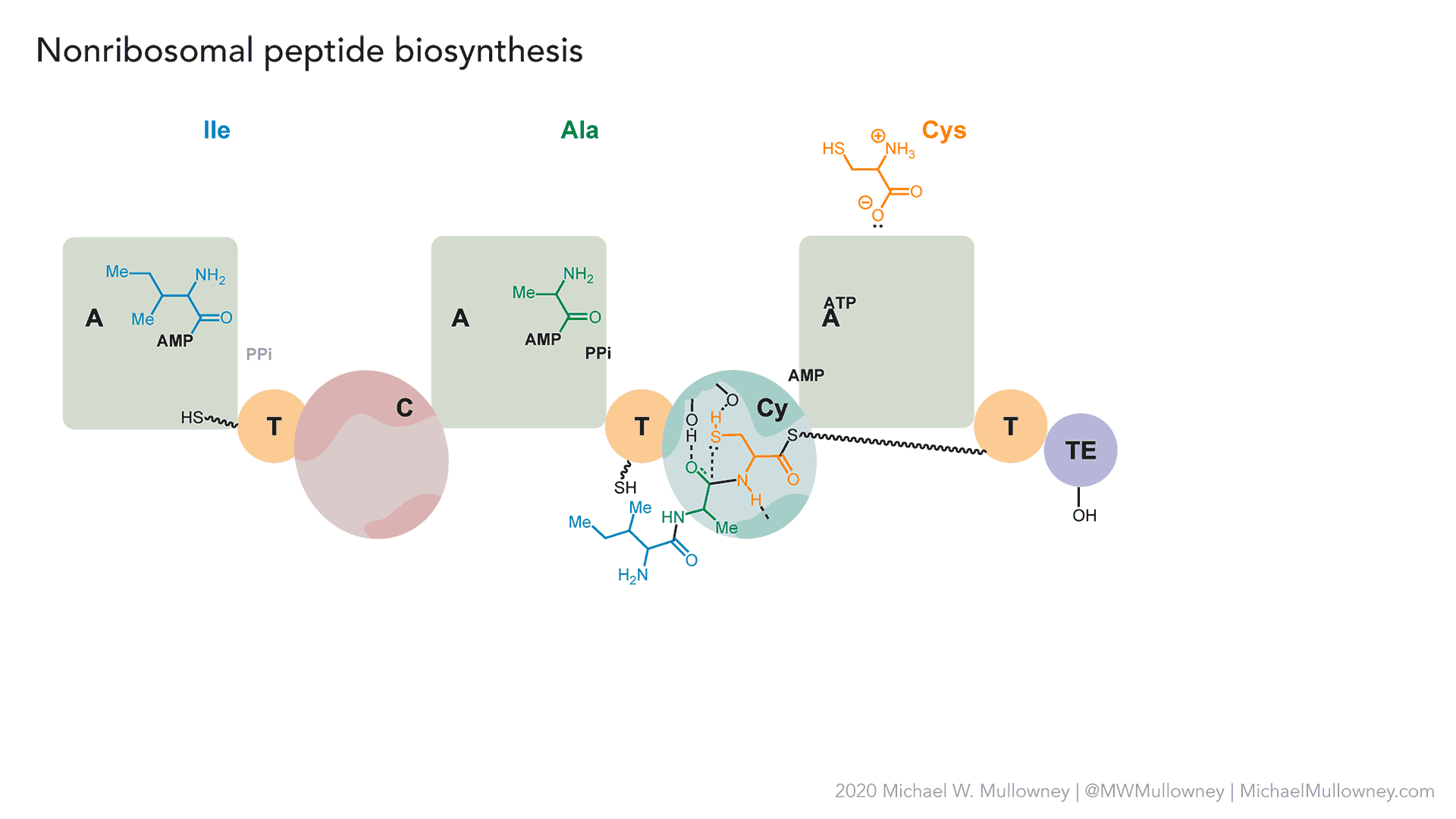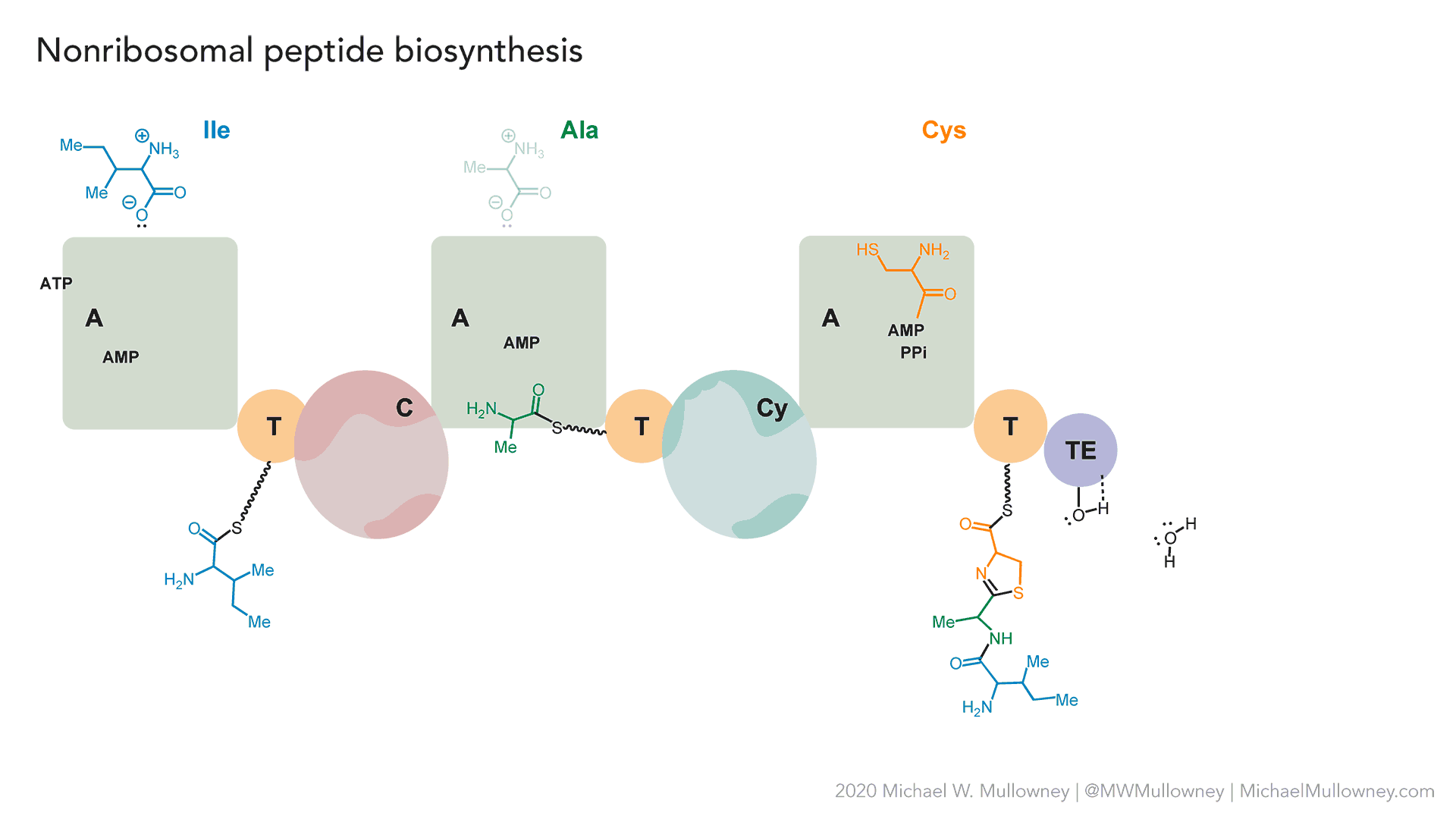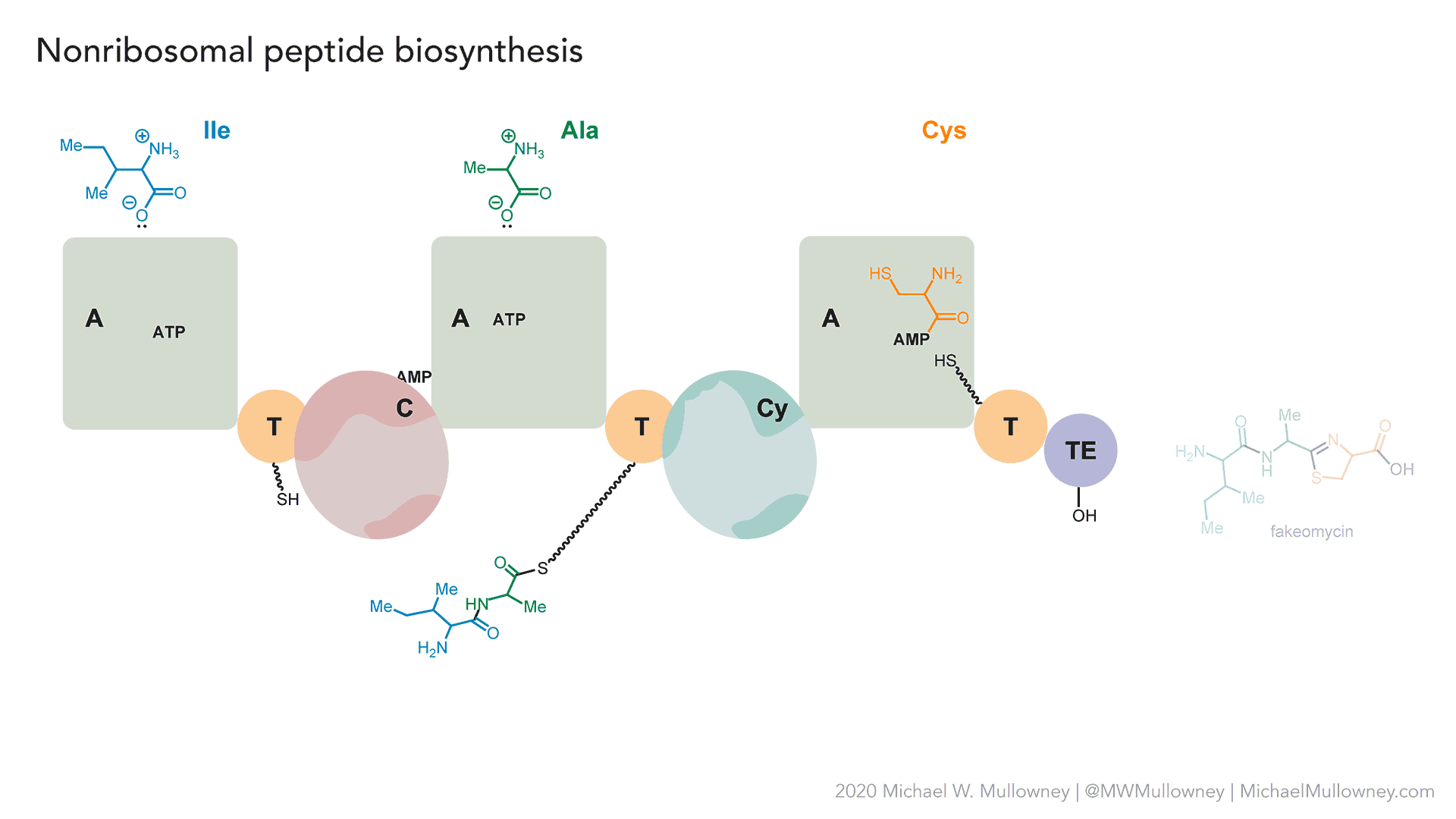
- A totally fake, yet plausible, NRPS biosynthesis of "fakeomycin" featuring a cyclization (Cy) domain. May, 2020.
Downloads:
Optimized for PowerPoint/Keynote/Slides or use however you like!
(right click, 'save link as...')
16:9 widescreen (1920x1080 px)
4:3 standard (1600x1200 px)
- Cover image artwork (and table of contents graphic) for Kafader, J.O.; Durbin, K.R.; Melani, R.D.; Des Soye B.J.; Schachner, L.F.; Senko, M.W.; Compton, P.D.; Kelleher, N.L. Individual Ion Mass Spectrometry Enhances the Sensitivity and Sequence Coverage of Top Down Mass Spectrometry. J. Proteome Res. 2020, 19, 1346–1350.
- Journal cover and media release artwork for Kafader, J.O.; Melani, R.D.; Durbin, K.R.; Ikwuagwu, B.; Early, B.P.; Fellers, R.T.; Beu, S.C.; Zabrouskov, V.; Makarov, A.A.; Maze, J.T.; Shinholt, D.L.; Yip, P.F.; Tullman-Ercek, D.; Senko, M.W.; Compton, P.D.; Kelleher, N.L. Multiplexed Single Ion Mass Spectrometry Improves Measurement of Proteoforms and Their Complexes. Nat. Methods. 2020,17, 391–394. Also featured in Phys.org, Drug Target Review, AZoOptics, Northwestern Now, and Northwestern Chemistry of Life Processes (CLP).
- Cover image artwork for Navarro-Muñoz, J.*; Selem-Mojica, N.*; Mullowney, M.W.*; Kautsar, S.; Tryon, J.H.; Parkinson, E.; Santos, E.D.L.; Yeong, M.; Cruz-Morales, P.; Abubucker, S.; Roeters, A.; Lokhorst, W.; Fernandez-Guerra, A.; Dias Cappelini, L.T.; Goering, A.W.; Thomson, R.J.; Metcalf, W.W.; Kelleher, N.L.; Barona-Gomez, F.; Medema, M.H. A computational framework to explore large-scale biosynthetic diversity. Nat. Chem. Biol. 2020, 16, 60–68.
*co-first authors
- Header image artwork for Nature Research Chemistry Community blog feature "BEHIND THE PAPER: Charting biosynthetic diversity in big data" by Marnix Medema
- Micromonospora sp. G039 cultured in the shape of the isopimara-2-one-3-ol-8,15-diene chemical structure. From: Mullowney, M.W.; Ó hAinmhire, E.; Tanouye, U.; Burdette, J.E.; Pham, V.C.; Murphy, B.T. A Pimarane Diterpene and Cytotoxic Angucyclines from a Marine-Derived Micromonospora sp. in Vietnam’s East Sea. Mar. Drugs. 2015, 13, 5815-27.
- Cover image artwork for Mullowney, M.W.; McClure, R.; Robey, M.; Kelleher, N.L..; Thomson, R.J. Natural products from biosynthetic assembly lines terminating in thioester reductases. Nat. Prod. Rep. 2018, 35, 847-78.

- A fractionated extract from fermentation of Streptomyces sp. F001, which contains diazaquinomycins A-F (red), and akashins (cyan). From: Mullowney, M.W.; Ó hAinmhire, E.; Shaikh, A.; Wei, X.; Tanouye, U.; Santarsiero, B.D.; Burdette, J.E.; Murphy, B.T. Diazaquinomycins E–G, novel diaza-anthracene analogs from a marine-derived Streptomyces sp. Mar. Drugs. 2014, 12, 3574-86.

- A fractionated extract from fermentation of Streptomyces sp. F001, which contains diazaquinomycins A-F (red), and akashins (cyan). From: Mullowney, M.W.; Ó hAinmhire, E.; Shaikh, A.; Wei, X.; Tanouye, U.; Santarsiero, B.D.; Burdette, J.E.; Murphy, B.T. Diazaquinomycins E–G, novel diaza-anthracene analogs from a marine-derived Streptomyces sp. Mar. Drugs. 2014, 12, 3574-86.

- Diving where the North American and Eurasian techtonic plates meet in Silfra, Iceland in 2014. Expedition covered for a feature in the Toronto Star. (L->R: Brian Murphy, Michael Mullowney)

- A round-bottom flask containing the cyan-colored akashin class of natural products from Streptomyces sp. F001. From: Mullowney, M.W.; Ó hAinmhire, E.; Shaikh, A.; Wei, X.; Tanouye, U.; Santarsiero, B.D.; Burdette, J.E.; Murphy, B.T. Diazaquinomycins E–G, novel diaza-anthracene analogs from a marine-derived Streptomyces sp. Mar. Drugs. 2014, 12, 3574-86.
- Round-bottom flasks containing the bright red-colored diazaquinomycin class of natural products from Streptomyces sp. F001. From: Mullowney, M.W.; Ó hAinmhire, E.; Shaikh, A.; Wei, X.; Tanouye, U.; Santarsiero, B.D.; Burdette, J.E.; Murphy, B.T. Diazaquinomycins E–G, novel diaza-anthracene analogs from a marine-derived Streptomyces sp. Mar. Drugs. 2014, 12, 3574-86.
- Round-bottom flasks containing the bright red-colored diazaquinomycin class of natural products from Streptomyces sp. F001. From: Mullowney, M.W.; Ó hAinmhire, E.; Shaikh, A.; Wei, X.; Tanouye, U.; Santarsiero, B.D.; Burdette, J.E.; Murphy, B.T. Diazaquinomycins E–G, novel diaza-anthracene analogs from a marine-derived Streptomyces sp. Mar. Drugs. 2014, 12, 3574-86.